However, OMVVs present complex manufacturing and regulatory issues 23. To broaden coverage, OMVVs containing multiple PorAs have been developed 19, 20, 21 and selected for the prevalence of PorA sequences in circulating strains 21, 22. 
The VR2 loop dominates PorA-specific immunity elicited by OMVVs, which offer limited or no cross-protection against strains expressing PorA with a different VR2 17, 18. Loops one and four are termed variable region 1 and 2 (VR1 and VR2), respectively, as they generate immune responses and are subject to antigenic variation 16. The immunodominant antigen in meningococcal OMVVs is PorA 14, an abundant outer-membrane porin with eight surface-exposed loops 15. OMVVs were first developed in the 1980s 11, 12, 13. meningitidis outer membrane vesicle vaccines (OMVV) and recombinant protein subunit vaccines. Two main approaches have been used to develop vaccines against serogroup B N. However, strategies for generating successful vaccines are hampered by pathogen diversity 9 and the difficulties associated with presenting epitopes from membrane-embedded surface proteins to the immune system 10. Given the rise in the emergence of multi-drug resistant bacteria 8, new approaches for vaccine development are required. meningitidis, for which a capsule-based vaccine is not feasible 7. pneumoniae 2, 3, un-encapsulated pathogens such as Neisseria gonorrhoeae and Moraxella catarrhalis 5, 6 and encapsulated serogroup B N. These pathogens include non-typeable strains of H. However, challenges remain in developing vaccines against pathogens for which toxoid and capsule-based vaccines are not feasible. Toxoid-based vaccines have almost eliminated diphtheria and tetanus in wealthy countries 1, while capsule-based vaccines have substantially reduced disease caused by Haemophilus influenzae 2, Streptococcus pneumoniae 3, and some strains of Neisseria meningitidis 4. Toxoid and capsule-based vaccines have prevented millions of deaths caused by bacterial pathogens. 
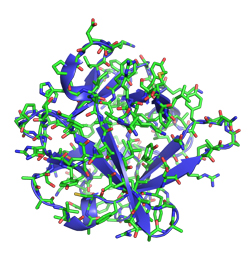
Application of ChAs is therefore a potentially powerful approach to develop multivalent subunit vaccines, which can be tailored to circumvent pathogen diversity. In mice, immunisation with ChAs generates fHbp and PorA antibodies that recognise the antigens expressed by clinical MenB isolates these antibody responses correlate with protection against meningococcal disease. Structural analyses demonstrate fHbp is correctly folded and the PorA VR2 epitope adopts an immunogenic conformation. MenB ChAs exploit factor H binding protein (fHbp) as a molecular scaffold to display the immunogenic VR2 epitope from the integral membrane protein PorA. 
ChAs are generated against serogroup B Neisseria meningitidis (MenB), the predominant cause of meningococcal disease in wealthy countries. Here we show how to use structure-based design to develop chimeric antigens (ChAs) for subunit vaccines. However, this is often hindered by antigenic diversity and difficulties encountered manufacturing membrane proteins. There is an urgent need to develop vaccines against pathogenic bacteria.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
